Using AI to control energy for indoor agriculture
30 September 2024
Published online 2 October 2011
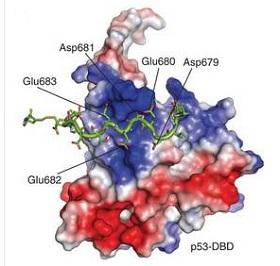
The heat shock protein Hsp90 — one of the highest proteins expressed in the cell — helps repress cancerous tumours by stabilizing the folded shape of the tumour suppressor protein p53. In this way, Hsp90 acts as a molecular chaperone, protecting p53 and enabling it to function.
Now, a team of researchers, including Horst Kessler of the King Abdulaziz University in Jeddah, Saudi Arabia, has figured out how Hsp90 protects the cell: by delicate interactions with a a certain domain of the p53 molecule.
Through a combination of NMR spectroscopy and several other biophysical methods, it was possible to determine the binding affinities between Hsp90 fragments and p53. For convenience, they used Hsp90 from yeast, previously shown to interact with p53 in a way analogous to human Hsp90.
The study revealed that the DNA-binding domain of p53 makes contact with each of the three distinct molecular domains of yeast Hsp90. p53 binds most strongly to the Hsp90 C-terminal domain, but it also associates weakly with binding sites in the middle and the N-terminal domains.
These results indicate that Hsp90's efficient chaperoning of p53 depends on a fine-tuned interplay of interactions with all three Hsp90 domains. Indeed, Kessler and colleagues found that p53 could be stabilized against thermal unfolding only when in contact with full-length Hsp90, suggesting that protection requires a concert of interactions with different Hsp90 domains.
doi:10.1038/nmiddleeast.2011.132
Stay connected: