Why We Need Arabic Language Models
24 August 2025
Published online 12 May 2011
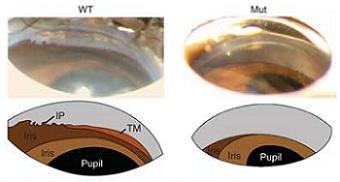
Angle-closure glaucoma affects about 16 million people worldwide, causing blindness in 4 million of them. This type of glaucoma results from an increase in intraocular pressure. It has long been thought that the iris obstructs the drainage networks of the eye, preventing the aqueous humour from leaving and thereby increasing the pressure. However, the exact genetic and molecular mechanisms of the condition are not yet clear.
Now, reporting in Nature Genetics, a team of researchers from Tunisia, Sweden and the United States has identified a genetic mutation that decreases the size of the eye, making it more likely that the iris will obstruct the angle where drainage occurs.
Mice carrying the mutation, named Grm4 mutants, had eyes with an average of 4.4% shorter axial length than those of normal mice, a reduction that occurred after birth. According to Simon John, one of the authors of the paper from the Howard Hughes Medical Institute, Maine, United States, this reduction was even more severe in some strains of mice.
The size of the lens, however, was similar in both groups of mice. Therefore, in the mutated mice, the lens occupied a larger proportion of the volume of the eye and so was more likely to obstruct the drainage systems. When the pupils dilate, the iris crowds in the angle of the eye. This causes an increase in angle closure in eyes susceptible to angle closure glaucoma. When the researchers dilated the pupils of the mice, intraocular pressure increased markedly in the mutant mice. This further supports the idea that the increase in intraocular pressure in these mice was related to angle closure in their eyes.
The researchers localized the Grm4 mutation to a gene encoding a serine protease called PRSS56. The Grm4 mutation did not affect the protease activity of PRSS56 on a synthetic substrate, but the researchers suggest that the mutation may alter its activity for endogenous substrates in the eye, leading to the changes that increase intraocular pressure.
The researchers also investigated six Tunisian families with posterior microphthalmia, a disease that is characterized by a reduction in the size of the posterior segment of the eye. They identified two mutations in PRSS56 in all of the humans studied who had the disease. These mutations resulted in PRSS56 having a similar defect to the Grm4 mutation.
Further research is needed to improve our understanding of PRSS56 and the role that mutations in this gene may have in other eye conditions.
doi:10.1038/nmiddleeast.2011.56
Stay connected: